Chemistry
Periodic Table
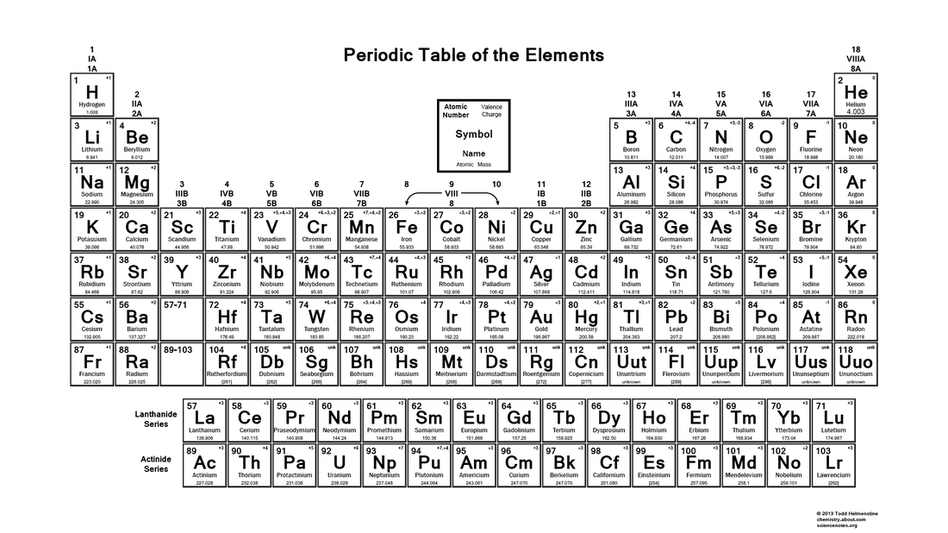
This right image on the left is the Periodic Table.
Periodic Table: a table of the chemical elements arranged in order of atomic number, usually in rows, so that elements with similar atomic structure appear in vertical columns.
The atomic number will be located in the upper left corner. It tells the number of protons and electrons.
Periodic Table: a table of the chemical elements arranged in order of atomic number, usually in rows, so that elements with similar atomic structure appear in vertical columns.
The atomic number will be located in the upper left corner. It tells the number of protons and electrons.
Models
There are two different models that you can make for an element.
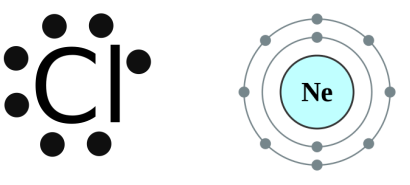
One is the Lewis Dot Structure which only give you the valence electrons.
And the other one is the Bohr Model. This shows every electron of the element.
One is the Lewis Dot Structure which only give you the valence electrons.
And the other one is the Bohr Model. This shows every electron of the element.
Lewis Dot Structure isn't difficult to do. All it is is the last ring of the Bohr Model. And you will just place the dots around the symbol.
molecule- the simplest form of a compound. A molecule is formed when two or more atoms combined.
Example) Water- h20 2 hydrogen 1 oxygen
compound- has to contain 2 elements. Which has to be different.
Example) h20- hydrogen and oxygen are different.
Example) Water- h20 2 hydrogen 1 oxygen
compound- has to contain 2 elements. Which has to be different.
Example) h20- hydrogen and oxygen are different.
Groups of the Perodic Table
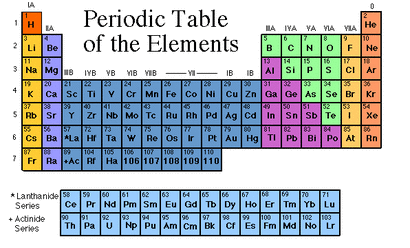
You can identify the groups by there location on the periodic table.
Dark Orange & Green- Non-Metals
Bright Yellow- Alkali Metals
Purple- " " Earth Metals
Dark Blue- Transional Metals
Pink- Metals
Light Yellow- Halogens
Light Orange- Noble Gases
LIght Blue- Rare Earth Metals (In 8th grade we didnt use these at all)
Determine number of protons, electrons, and neutrons.
You can read the atomic number and it will tell you how many electrons and protrons that element has.
To find the number of neutrons you would round the atomic mass to the nearest whole. Then you subtract the atomic mass from the atomic number. (As also seen in the video above about Bohr Models)
To find the number of neutrons you would round the atomic mass to the nearest whole. Then you subtract the atomic mass from the atomic number. (As also seen in the video above about Bohr Models)
Easier ways
There is an easier way to find the valence electrons. If you count the rows across the top from left to right the last digit of the number you get is the number of valence electrons.
Why are elements in groups and periods?
Elements are grouped up into groups because they'll have the same number of electrons in their outer shell and will behave similar chemically. ************
Mass and Volume
Volume IMPORTANT
- Measured in meters^3 VOLUME=MASS/DENSITY
- l * w * h which means
- 1 in^3= 16.387064 cm^3 MASS=VOLUME*DENSITY
- 1 cm^3= 1 ml
Mass
- Measured in grams
-responsible for the weight of the object
- Measured in meters^3 VOLUME=MASS/DENSITY
- l * w * h which means
- 1 in^3= 16.387064 cm^3 MASS=VOLUME*DENSITY
- 1 cm^3= 1 ml
Mass
- Measured in grams
-responsible for the weight of the object
Physical vs Chemical Change
Physical and chemical change are very different from each other.
A physical property is one you can change without changing the identity of the substance.
Physical Changes- color, smell, and freezing point
Phase changes are physical changes because even though it has changed it still has the same chemical things that it had before the change.
Evaportation is also a physical change because even though the might be evaporated it still has the same chemicall form as before.
A chemical property is one that can only be observed when the indentity of the substance is changed.
Chemical Changes- burning, exploding, rust, and baking a cake
Signs of Chemical Change- color change, release of a gas, formation of a solid, production of heat or light, or substance dissolving
A physical property is one you can change without changing the identity of the substance.
Physical Changes- color, smell, and freezing point
Phase changes are physical changes because even though it has changed it still has the same chemical things that it had before the change.
Evaportation is also a physical change because even though the might be evaporated it still has the same chemicall form as before.
A chemical property is one that can only be observed when the indentity of the substance is changed.
Chemical Changes- burning, exploding, rust, and baking a cake
Signs of Chemical Change- color change, release of a gas, formation of a solid, production of heat or light, or substance dissolving
Balancing Equations
To balance a chemical equation, you have to multiply or divide each side by a number to make the sides equal each other.
To help, here is a video by Khan Academy about balancing equations that is very more detailed!
To help, here is a video by Khan Academy about balancing equations that is very more detailed!